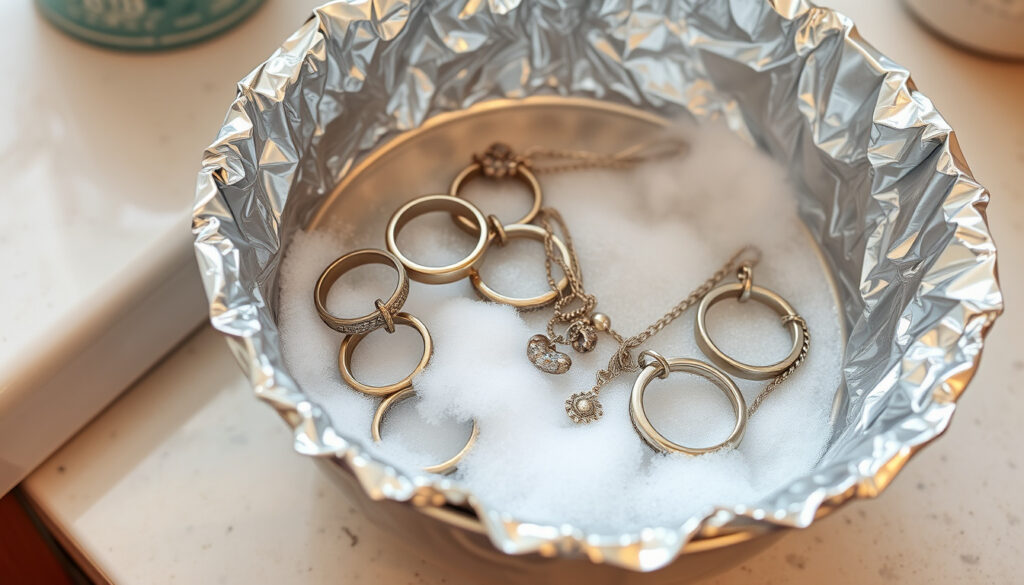
How to Clean Silver Jewelry – Baking Soda and Foil Methods
How to Clean Silver Jewelry at Home
Silver jewelry develops tarnish over time due to exposure to sulfur compounds in the air. This natural process affects even the finest sterling silver pieces, leaving them dull and discolored. Fortunately, effective cleaning methods using common household items can restore shine without damaging the metal.
The most popular home remedies involve baking soda, aluminum foil, and simple dish soap. These methods work through chemical reactions that lift tarnish without abrasive polishing, making them safe for delicate jewelry. Understanding which technique suits your specific pieces ensures optimal results while protecting your investment.
This guide covers the complete range of home cleaning options, from gentle soap treatments to the powerful electrolytic reaction method preferred by museum conservators. Each approach addresses different levels of tarnish and types of silver jewelry.
Materials: Foil, baking soda, salt, hot water
Time: 2-30 minutes
Best for: Heavy tarnish, necklaces
Materials: Non-gel toothpaste, soft cloth
Time: 1-5 minutes
Best for: Quick spot cleaning
Materials: Baking soda, water
Time: 30 seconds-10 minutes
Best for: Light tarnish, crevices
Materials: Dish soap, warm water
Time: 5-10 minutes
Best for: Regular maintenance
- The baking soda and aluminum foil method triggers an electrolytic chemical reaction that transfers tarnish (silver sulfide) from the silver to the foil
- This non-abrasive approach preserves silver layers and is the same technique used by museum conservators for valuable pieces
- Sterling silver responds particularly well to electrolytic cleaning, often achieving “virtually new” results on heavily tarnished items
- Never use abrasive polishes on plated or thin silver items, as they wear away metal layers
- Pearls and certain gems are sensitive to soaking methods and should be removed before cleaning
- Commercial polishes contain toxic chemicals and accelerate silver wear—home remedies are safer and more eco-friendly
- Storing cleaned silver in anti-tarnish bags prevents recurrence by blocking sulfur exposure
| Aspect | Details |
|---|---|
| Tarnish cause | Reaction between silver and hydrogen sulfide in air |
| Safe methods | Baking soda, foil, dish soap, microfiber polishing |
| Avoid | Commercial polishes, abrasive materials, prolonged soaking on gems |
| Frequency | Monthly for frequently worn pieces |
| Gemstone caution | Remove pearls, opals, and heat-sensitive gems before cleaning |
| Sterling silver | Safe for electrolytic method; contains 92.5% silver |
| Preventive storage | Anti-tarnish bags limit sulfur exposure |
| Safety for silver | Highest: foil method; Moderate: paste; Low: commercial polishes |
How to Clean Tarnished Silver Jewelry
Tarnish appears when silver reacts with sulfur compounds, forming silver sulfide on the surface. This dark coating starts as a yellowish tint and progresses to brown and eventually black. Heavily tarnished pieces require stronger intervention than regular maintenance cleaning.
Understanding Tarnish Severity
Light tarnish appears as a slight dullness or yellowish hue that dulls the metal’s shine. Moderate tarnish shows brownish patches and noticeably reduced reflectivity. Heavy tarnish creates black discoloration that may obscure details on intricate pieces. The cleaning method should match the severity—light tarnish responds to gentle soap or baking soda paste, while heavy tarnish typically requires the foil and baking soda electrolytic reaction.
Regular wear actually slows tarnish buildup because skin oils provide a mild protective barrier. Jewelry stored in humid environments tarnishes faster due to increased sulfur activity. Pieces kept in anti-tarnish bags or lined jewelry boxes maintain their shine longer between cleanings.
When to Choose Each Method
Daily wear items benefit from quick soap and water cleaning to remove body oils and prevent buildup. Weekly or monthly deeper cleaning with baking soda paste addresses accumulated tarnish without harsh treatment. Heavily tarnished heirloom pieces or vintage finds respond best to the full foil and baking soda soak method, which can dramatically restore appearance in minutes.
For frequently worn pieces, a quick wipe with a microfiber cloth after wearing removes oils and sweat that contribute to tarnish formation. This simple habit extends the time between thorough cleanings.
Clean Silver Jewelry with Baking Soda and Aluminum Foil
The baking soda and aluminum foil method represents the gold standard for home silver cleaning. This technique exploits basic electrochemistry—tarnish transfers from silver to aluminum through a saltwater solution, leaving the silver clean without scrubbing or chemical polish.
The Science Behind the Method
When silver sulfide contacts aluminum in a hot saline solution, a galvanic cell forms. Electrons flow from the aluminum (more reactive metal) to the silver sulfide, reducing it back to metallic silver while the aluminum oxidizes. The tarnish literally migrates through the solution and attaches to the foil, which darkens as it collects silver sulfide.
This reaction only occurs when silver and foil make direct contact through the electrolyte solution. The jewelry must touch the foil surface for the electrical circuit to complete. The water temperature affects reaction speed—hotter solutions work faster but require careful handling.
Basic Soak Method for Necklaces and Chains
Line a glass dish or pan with aluminum foil, shiny side facing up. Dissolve 2 tablespoons of baking soda and 2 tablespoons of salt in 2 cups of boiling water. Carefully place your silver jewelry on the foil, ensuring pieces touch the surface without overlapping. Allow the items to soak for 2 to 15 minutes depending on tarnish severity—the water will gradually turn yellowish as tarnish releases.
Remove pieces using kitchen tongs (metal tongs may conduct heat, so use plastic or silicone alternatives). Rinse each item in warm soapy water to remove any residue, then dry thoroughly with a soft microfiber cloth. Buff gently to restore full shine. Heavily tarnished pieces may require a second treatment.
Boiling Variation for Stubborn Tarnish
For very dark or stubborn tarnish, bring the cleaning process to a boil. Add your foil-lined items to a pot containing 2-3 inches of water, along with 1 teaspoon of baking soda and 1 teaspoon of salt per quart of water. Bring to a gentle boil for approximately 4 minutes before removing and rinsing as usual. This accelerated reaction handles years of accumulated tarnish in a single session.
Foil-Wrap Technique for Delicate Pieces
Intricate jewelry with crevices, chains, or multiple components benefits from individual wrapping. Place each piece in its own sheet of aluminum foil, shiny side facing inward. Submerge the wrapped items in hot water containing 1 cup of baking soda per gallon. Allow soaking for up to 30 minutes, then unwrap, rinse, and dry. This method ensures the electrolytic reaction reaches all surface areas.
Results from the foil method prove dramatic on sterling silver. Jewelry transforms from blackened or heavily tarnished to bright and reflective, often appearing nearly new. The technique works safely on gold, brass, and copper as well, though results vary depending on the metal’s condition and composition.
Best Home Remedies and Alternatives for Cleaning Silver
Several alternatives exist for situations where baking soda, foil, or specific supplies are unavailable. Each method carries distinct advantages and limitations worth understanding before proceeding.
Toothpaste as Silver Polish
Standard non-gel toothpaste serves as a mild abrasive polish for silver. The method involves applying a small amount directly to a soft cloth or sponge and rubbing the tarnished area gently. Five to six strokes per section typically suffice for light tarnish. Rinse thoroughly to remove all toothpaste residue, as the mild abrasives can leave deposits in crevices if not washed away completely.
Toothpaste works through mechanical abrasion rather than chemical reaction, making it less effective for heavy tarnish removal. Repeated use on the same areas may create micro-scratches that accumulate over time, dulling the surface eventually. This approach suits occasional spot cleaning rather than comprehensive restoration. Avoid using whitening toothpastes or those containing baking soda, as these prove more abrasive.
Baking Soda Paste Without Foil
Mix baking soda with just enough water to form a thick paste consistency. Apply this mixture using a soft sponge or cloth, working into crevices and detailed areas. Allow the paste to remain for 30 seconds to 10 minutes depending on tarnish severity, then rinse with warm water and dry immediately. This method reaches intricate details that soaking methods might miss.
The paste approach requires gentle rubbing, unlike the chemical reaction of the foil method. For best results, use a soft-bristled baby toothbrush on detailed areas. This technique works well for lightly tarnished items and regular maintenance between deeper cleanings. It proves particularly useful for jewelry with embossed patterns or filigree work.
Commercial Cleaners: When to Avoid
Commercial silver polishes and dips remain available but carry significant drawbacks. These products typically contain toxic chemicals requiring protective gloves and adequate ventilation. More critically, repeated use wears down silver layers, shortening the lifespan of valuable pieces. Abrasion-based polishes remove metal along with tarnish, thinning delicate items with each application.
Museum conservators and professional jewelers consistently favor non-abrasive electrochemical methods precisely because they preserve precious metal. Home remedies achieve comparable results without health risks or material degradation. For these reasons, commercial products should be considered a last resort rather than a primary cleaning approach.
The foil and baking soda electrolytic method scores highest for silver preservation, effectiveness on heavy tarnish, and safety for detailed jewelry. Toothpaste offers quick results but risks surface damage over time. Soap and water excel for maintenance cleaning but cannot address established tarnish.
Never submerge jewelry containing pearls, opals, amber, or other organic or heat-sensitive gems in hot water or cleaning solutions. These materials absorb liquids, crack under temperature changes, or may become loose from their settings. Remove all stones before cleaning and clean them separately with appropriate methods.
Cleaning Silver Necklaces and Delicate Pieces
Necklaces present unique challenges due to their chain links and tendency to tangle. The foil method works exceptionally well for chains when pieces make direct contact with the aluminum surface. Lay chains flat on the foil, ensuring no overlapping sections, as overlapping prevents proper electrical contact and reduces cleaning effectiveness.
For necklaces with pendant stones, remove the pendant before cleaning if possible. Clean the chain using the standard foil soak, then address the pendant separately using a baking soda paste applied with a soft cloth. This approach protects stone settings from thermal shock and prevents moisture damage to porous materials.
Testing Before Full Treatment
Before applying any cleaning method to valuable or sentimental pieces, test on a small, less visible area first. This precaution identifies potential adverse reactions, particularly with plated items where the thin silver layer may behave differently than solid sterling silver. A test also confirms that stones and settings will withstand the cleaning process without damage.
If uncertainty exists about a piece’s composition—whether it is solid sterling, plated, or a silver-colored alloy—consult a professional jeweler before attempting home cleaning. Plated items respond poorly to electrolytic cleaning and may strip entirely if the base metal is more reactive than aluminum.
After cleaning, store silver jewelry in anti-tarnish bags or cloth-lined boxes. These protective containers limit sulfur exposure from the air, significantly extending the time between cleanings. Adding anti-tarnish strips to jewelry boxes provides additional protection for larger collections.
What Works and What Remains Uncertain
Scientific understanding of silver tarnish and its removal is well-established, though practical application involves some variables worth noting.
| Established Information | Variables and Uncertainties |
|---|---|
| Baking soda and foil creates electrolytic reaction | Optimal soaking time varies by tarnish severity |
| Hot water accelerates the cleaning process | Temperature tolerance of specific gemstones |
| Abrasion damages silver over time | Results on unknown plated items unpredictable |
| Pearls damaged by soaking methods | Exact effectiveness varies by silver purity |
| Anti-tarnish storage prevents recurrence | Long-term effects of repeated cleaning unknown |
Why Silver Tarnishes and Prevention Basics
Silver tarnishes through a natural chemical process called oxidation, though technically the reaction involves sulfur compounds rather than oxygen. Silver sulfide forms when metallic silver reacts with hydrogen sulfide present in the air, cosmetics, and certain foods. This reaction occurs more rapidly in humid environments and near rubber, cardboard, and certain fabrics that release sulfur compounds.
Sterling silver, an alloy containing 92.5% pure silver and 7.5% other metals (usually copper), tarnishes more readily than pure silver. The copper component accelerates tarnish formation through its own oxidation reactions. Despite this vulnerability, sterling silver’s durability makes it the preferred choice for quality jewelry that withstands daily wear.
Prevention strategies focus on limiting sulfur exposure and moisture contact. Removing jewelry before swimming, showering, or applying cosmetics reduces exposure to corrosive substances. Placing silica gel packets in jewelry boxes absorbs ambient moisture. Regular wearing actually helps maintain shine because skin oils provide a temporary protective coating against tarnish development.
Expert Methods and Professional Approaches
Museum conservators rely on electrolytic cleaning methods for valuable silver artifacts precisely because these techniques remove tarnish without removing metal. The baking soda and foil approach mirrors professional restoration while remaining safe for home use.
Based on methods documented by conservation professionals
Professional jewelers employ specialized equipment including ultrasonic cleaners, steam cleaners, and proprietary polishing compounds calibrated for specific silver types. These methods achieve superior results but risk damage if misapplied. Ultrasonic cleaners vibrate items at high frequencies to dislodge debris but can loosen stones from settings. Steam cleaning provides chemical-free shine restoration but requires training to prevent burns and handle thermal shock properly.
For valuable or intricate pieces, annual professional cleaning ensures proper care while identifying potential issues like worn clasps, loose stones, or damaged chains before they result in loss. Many jewelers offer complimentary cleaning with purchase, making professional maintenance accessible for most budgets.
Summary and Next Steps
Cleaning silver jewelry at home requires matching the method to the piece and tarnish severity. The baking soda and aluminum foil electrolytic reaction offers the most effective, safe, and eco-friendly approach for most tarnished silver items. Soap and water work well for maintenance cleaning, while toothpaste provides occasional spot treatment with awareness of its abrasive limitations. Avoid commercial polishes and abrasive materials that damage silver over time.
Remove all gemstones—especially pearls, opals, and amber—before applying any wet cleaning method. Test unfamiliar pieces in an inconspicuous area and store cleaned items in anti-tarnish bags to prevent quick recurrence. With proper care and regular maintenance, quality silver jewelry maintains its beauty for generations. For those interested in related topics, the Washing Up Liquid Environmental Impact article explores sustainable cleaning alternatives that complement these silver care techniques.
Frequently Asked Questions
How do you clean sterling silver?
Clean sterling silver using the baking soda and aluminum foil method for best results. Line a container with foil, add hot water with 2 tablespoons each of baking soda and salt per cup of water, soak for 2-15 minutes until tarnish lifts, then rinse and dry. This electrolytic method removes tarnish without abrasion, preserving the metal.
How do you clean a silver necklace?
Remove any pendants with stones first. Lay the chain flat on aluminum foil in a glass dish, ensuring no overlapping. Pour boiling solution of baking soda and salt over the chain, soak 2-10 minutes until the water turns yellowish. Remove with tongs, rinse in warm soapy water, dry, and buff with microfiber cloth.
How do you remove tarnish from sterling silver without baking soda?
Use non-gel toothpaste applied with a soft cloth, rubbing gently for 5-6 strokes per area. Alternatively, use baking soda paste (baking soda mixed with water) applied with a sponge. Both methods involve mild abrasion and work best for light to moderate tarnish. For heavy tarnish, baking soda remains the most effective home option.
What is the best silver cleaner?
The best silver cleaner for home use is a baking soda and aluminum foil solution, which provides electrochemical tarnish removal without abrasives or toxic chemicals. This method is preferred by museums and professional conservators because it removes only tarnish, preserving precious silver layers. Commercial polishes should be avoided due to toxicity and material wear.
How do you clean silver without aluminum foil?
Mix baking soda with water to form a paste, apply with a soft cloth or sponge, wait 30 seconds to 10 minutes, then rinse and dry. Alternatively, use non-gel toothpaste as a mild polish. These methods work through gentle abrasion rather than chemical reaction, so they require more rubbing and may be less effective on heavy tarnish.
Can you use toothpaste to clean silver jewelry?
Yes, non-gel toothpaste works as a mild silver polish. Apply a small amount to a soft cloth, rub gently for 5-6 strokes, rinse thoroughly, and dry. This method works for light tarnish but risks micro-scratches with repeated use. Avoid whitening toothpastes or using this method on valuable or heavily plated items.
How do you clean silver jewelry that has turned black?
Black tarnish indicates heavy silver sulfide buildup requiring the baking soda and foil electrolytic method. Boil a solution of baking soda and salt, line a container with foil, soak for up to 15 minutes, and repeat if necessary. The electrolytic reaction reverses the tarnishing process, returning black silver to its original shine.
How often should you clean silver jewelry?
Clean frequently worn silver jewelry monthly with baking soda paste or the foil method. Remove tarnish as soon as it appears rather than allowing it to build up heavily. Between cleanings, wipe pieces with a microfiber cloth after wearing to remove oils and prevent rapid tarnish development.








